 Use this ISO 13485 checklist to determine whether your company’s QMS meets the ISO standards and take corrective actions. A QMS will typically include processes and procedures for analyzing the quality of products and services, managing risk related to quality issues, maintaining records of performance (including corrective actions), and training employees on how to use the system. An ISO13485 audit checklist can be used as part of this process. Internal Audit Checklist ISO 13485 templates ISO 13485 / MDR document template: Internal Audit Checklist. To prepare for an ISO 13487 auditing process, it is important to have baseline information about your organization’s QMs already in place. You will need to have copies of the ISO 13485:2016 standard and Part 820, quality system regulation / code of federal regulations (21 CFR 820) to use along with this checklist. This provides assurance that your organization meets global safety and quality standards so that you can maintain customers’ trust and protect your business reputation. ©2016 13485 Store of 67 The ISO 13485:2016 / FDA-CFR Internal Audit Checklist This list has been prepared for you by the 13485 Store. A certification audit involves conducting an in-depth evaluation of a company’s QMS to determine its compliance with specific standards. ISO 13485 requirements checklists help assess whether a company is ready to undergo an ISO 13485 certification audit by a third party. 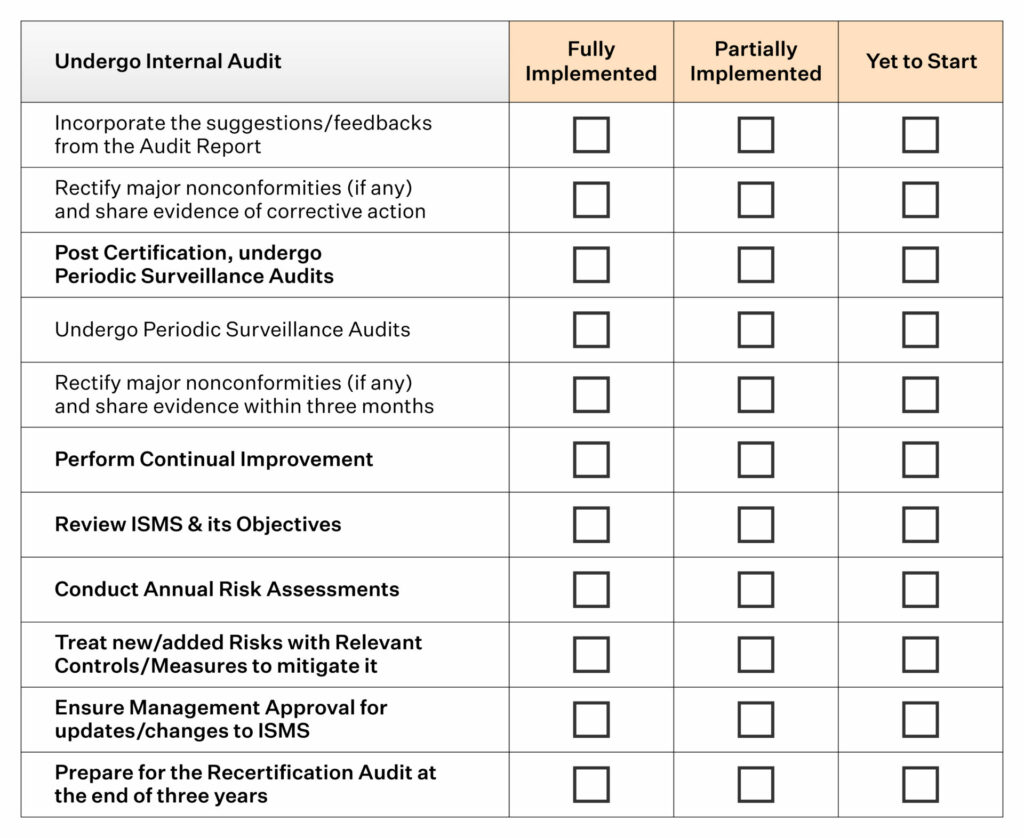
The ISO 13485:2016 standard establishes a framework for the quality management system (QMS) requirements for healthcare products and services. 
It’s important for businesses to be compliant with these standards because it shows they are taking their quality management seriously. ISO 13485 is a set of international standards that help organizations identify, control, and prevent quality problems. Make your voice heard in our 2024 global quality survey. Use this checklist to ensure compliance with the particular and unique areas of the standard. ISO 134 + Additional Requirements (and missing a few ISO 9001 requirements. The checklist provides questions that refer to the ISO 13485: 2003 standard. 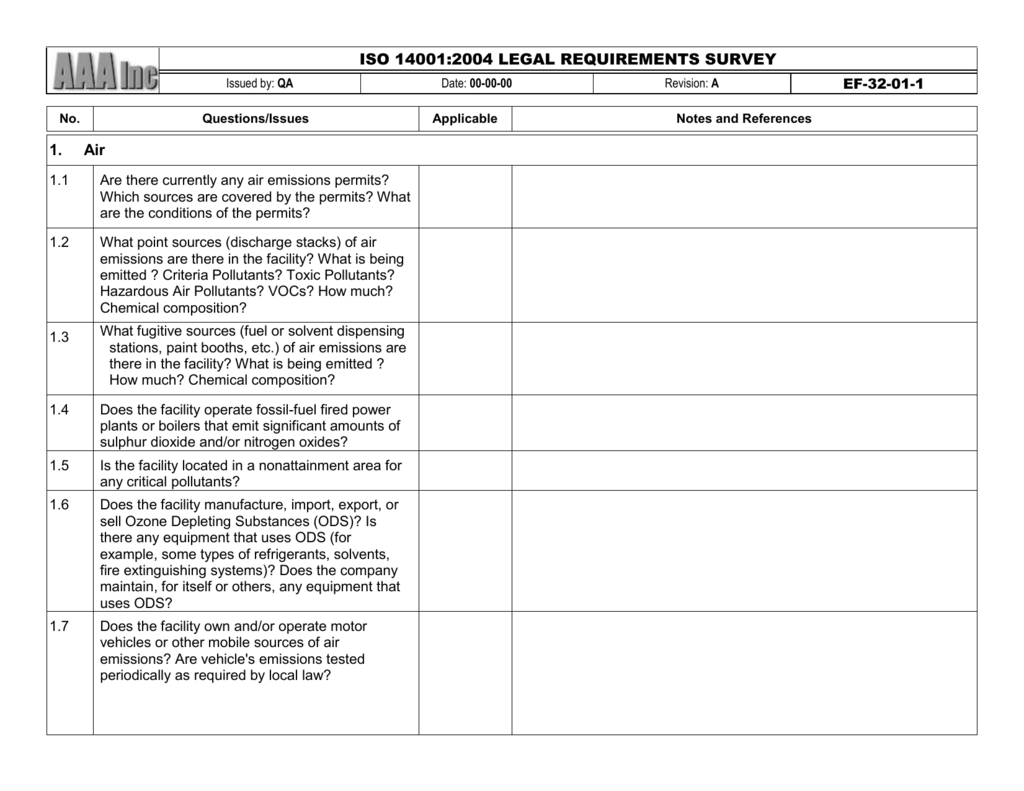
(Please note that some of the documents will not be mandatory if the company does not perform relevant processes related to ISO 13485 documentation requirements): Standard clause 4. ISO 13485 lays out the broad quality requirements for the modern medical device quality management system. ISO 13485:2003/FDA QSR - INTERNAL AUDIT CHECKLIST. Inspect your quality management system with an ISO 13485 audit checklist Here are the required ISO 13485 documents, categorized according to the clauses of the standard. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
